New research highlights an unexpected pathway linking cellular stress to blood system decline
As we age, stem cells responsible for maintaining our blood and immune systems gradually lose their resilience, but why this happens has remained unclear. A novel study from Japan and the US turns the spotlight on MLKL, a protein traditionally associated with cell death, uncovering an unexpected new dimension to its role. The findings shed light on how cellular stress shapes stem cell aging and highlight potential pathways for developing therapies to counter age-related decline.
As we age, our ability to maintain healthy blood and a strong immune system gradually declines, largely because hematopoietic stem cells (HSCs), the cells responsible for producing all blood cell types, begin to lose their effectiveness. Normally, HSCs can both self-renew and generate a balanced mix of blood cells, but over time they produce fewer new cells, favor certain cells such as myeloid cells over lymphoid cells, and struggle to support a robust immune response. Accumulated cellular damage, shifts in gene activity, ongoing low-level inflammation, and changes in the bone marrow environment, all appear to contribute to this decline. However, the precise mechanisms by which these diverse stresses converge to weaken HSCs have remained unclear.
Researchers from The University of Tokyo, Japan, and St. Jude Children’s Research Hospital, USA, sought to uncover a mechanism explaining how age-related stresses drive HSC functional deterioration, focusing on the receptor-interacting protein kinase 3 (RIPK3)-mixed lineage kinase like (MLKL) signaling axis—a pathway traditionally associated with necroptosis, or programmed cell death. The study was led by Dr. Masayuki Yamashita, an Assistant Member at St. Jude Children’s Research Hospital, who, at the time of the investigation, was an Assistant Professor at The Institute of Medical Science, The University of Tokyo. The other co-authors include Dr. Atsushi Iwama from The Institute of Medical Science, The University of Tokyo, and Dr. Yuta Yamada from St. Jude Children’s Research Hospital, who was a graduate student at The Institute of Medical Science, The University of Tokyo.
Explaining the motivation behind the study, Dr. Yamashita says, “We discovered an unexpected phenotype in HSCs of MLKL-knockout mice repeatedly treated with 5-fluorouracil, where aging-associated functional changes were markedly attenuated despite no detectable difference in HSC death, prompting us to investigate whether this pathway might induce functional changes beyond cell death.” This observation shifted the research focus toward a non-lethal role of MLKL—a concept later highlighted in their study, published in Volume 17 of the journal Nature Communications on April 6, 2026.
To investigate this, the team employed a combination of genetic mouse models, stress treatments, and functional assays. They used wild-type, MLKL-deficient, and RIPK3-deficient mice, along with specialized reporter mice capable of detecting MLKL activation through a Förster resonance energy transfer-based biosensor. Mice were exposed to stressors mimicking aging, including inflammation, replication stress, and oncogenic stress. HSC function was then assessed primarily through bone marrow transplantation, which measures the ability of stem cells to regenerate the blood system. Complementary analyses included flow cytometry, ex vivo expansion, RNA-seq, assay for transposase-accessible chromatin-seq, high-resolution microscopy, metabolic assays, and mitochondrial analyses, enabling a detailed understanding of how non-lethal MLKL activation impairs HSC function at molecular, cellular, and organelle levels.
The results revealed a novel, non-necroptotic role for MLKL in HSC aging. While MLKL is typically linked to cell death, its activation in HSCs did not increase cell death or reduce cell numbers. Instead, stress-induced MLKL activation was transient and localized to mitochondria, where it caused direct damage, reducing membrane potential, altering mitochondrial structure, and impairing energy production. These changes led HSCs to exhibit hallmark features of aging, such as diminished self-renewal, reduced lymphoid differentiation, and a shift toward myeloid-biased output.
Crucially, deletion or inactivation of MLKL significantly alleviated these defects. MLKL-deficient HSCs maintained regenerative capacity, produced healthier immune cells, displayed lower DNA damage, and preserved mitochondrial function, even under stress or in aged animals. Interestingly, these improvements occurred without substantial changes in gene expression or chromatin accessibility, suggesting that MLKL drives HSC aging primarily through post-transcriptional and organelle-level mechanisms, rather than through transcriptional regulation or inflammation.
These findings have broad implications for understanding aging and potential therapies. By linking diverse stress signals to mitochondrial dysfunction via MLKL, the study identifies a common pathway underlying HSC aging. Dr. Yamashita emphasizes, “In the longer term, this research could lead to therapies that preserve the function of hematopoietic stem cells, ultimately improving recovery and long-term health for patients undergoing chemotherapy, radiation, or transplantation. By revealing how non-lethal activation of cell-death pathways drives stem cell aging, these findings may inspire new classes of mitochondrial-protective or necroptosis-modulating drugs.”
In conclusion, this study uncovers a previously unrecognized role of MLKL as a non-lethal regulator of stem cell aging. Rather than inducing cell death, MLKL acts as a stress-responsive factor that damages mitochondria and drives functional decline in HSCs. These insights not only redefine the role of necroptosis-related proteins but also open new avenues for understanding and potentially intervening in the aging of the hematopoietic system.
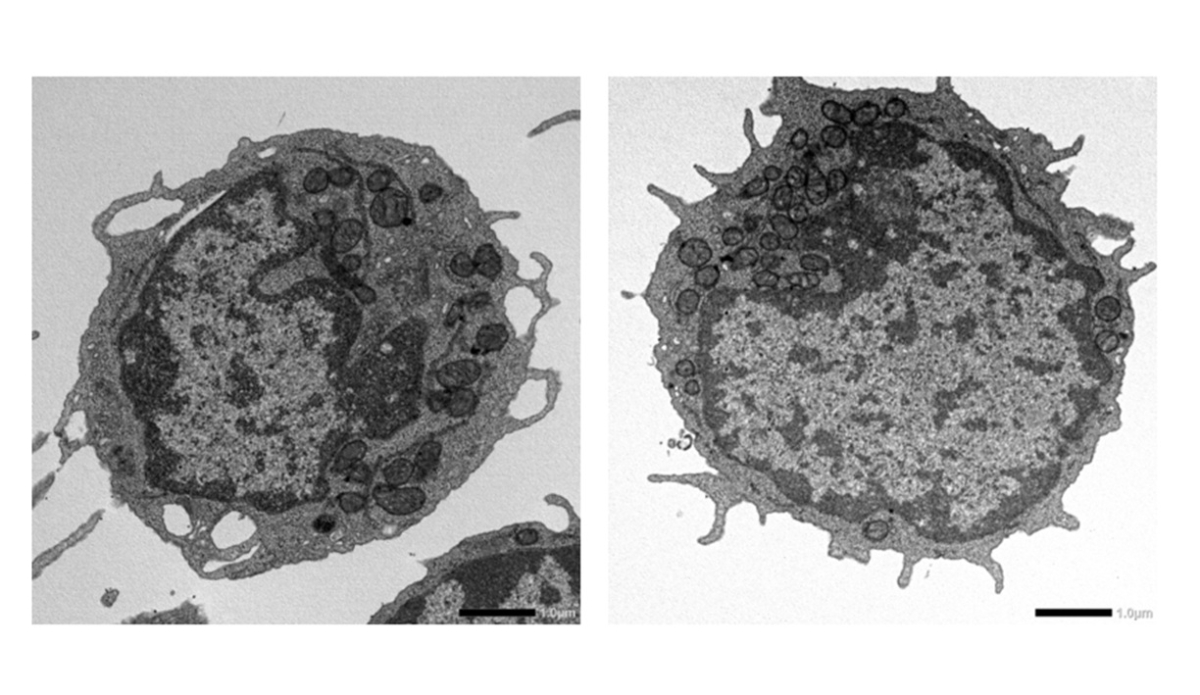
Transmission electron microscopy images of hematopoietic stem cells from 18‑month‑old wild‑type (left) and MLKL‑deficient (right) mice. The images were captured by Dr. Yuta Yamada and Dr. Masayuki Yamashita in collaboration with Dr. Yuji Watanabe and Dr. Hiroshi Sagara at Medical Proteomics Laboratory, The Institute of Medical Science, The University of Tokyo, Japan.
Reference
Journal:Nature Communications
Title of original paper:
Non-necroptotic MLKL function damages mitochondria and promotes hematopoietic stem cell aging
DOI:
10.1038/s41467-026-71060-4
Authors:
Yuta Yamada1,2, Jinjing Yang2, Akiho Saiki-Tsuchiya2, Yuji Watanabe3, Shuhei Koide2, Shin Murai4, Yuriko Sorimachi5, Yu Fukuda1, Kenta Sumiyama6, Hiroshi Sagara3, Hiroyasu Nakano7,8, Keiyo Takubo5,9, Atsushi Iwama2,10, and Masayuki Yamashita1,2
Affiliations:
1Division of Experimental Hematology, Department of Hematology, St. Jude Children’s Research Hospital, USA
2Division of Stem Cell and Molecular Medicine, Centre for Stem Cell Biology and Regenerative Medicine, The Institute of Medical Science, The University of Tokyo, Japan
3Medical Proteomics Laboratory, The Institute of Medical Science, The University of Tokyo, Japan
4Department of Biochemistry, Faculty of Medicine, Toho University, Japan
5Department of Stem Cell Biology, National Institute of Global Health and Medicine, Japan Institute for Health Security, Japan
6Department of Animal Sciences, Graduate School of Bioagricultural Sciences, Nagoya University, Japan
7Unit of Host Defense, Faculty of Medicine, Toho University, Japan
8Research Administration Organization, Toho University, Japan
9Department of Cell Fate Biology and Stem Cell Medicine, Tohoku University Graduate School of Medicine, Japan
10Laboratory of Cellular and Molecular Chemistry, Graduate School of Pharmaceutical Sciences, The University of Tokyo, Japan
2Division of Stem Cell and Molecular Medicine, Centre for Stem Cell Biology and Regenerative Medicine, The Institute of Medical Science, The University of Tokyo, Japan
3Medical Proteomics Laboratory, The Institute of Medical Science, The University of Tokyo, Japan
4Department of Biochemistry, Faculty of Medicine, Toho University, Japan
5Department of Stem Cell Biology, National Institute of Global Health and Medicine, Japan Institute for Health Security, Japan
6Department of Animal Sciences, Graduate School of Bioagricultural Sciences, Nagoya University, Japan
7Unit of Host Defense, Faculty of Medicine, Toho University, Japan
8Research Administration Organization, Toho University, Japan
9Department of Cell Fate Biology and Stem Cell Medicine, Tohoku University Graduate School of Medicine, Japan
10Laboratory of Cellular and Molecular Chemistry, Graduate School of Pharmaceutical Sciences, The University of Tokyo, Japan
About The Institute of Medical Science, The University of Tokyo
The Institute of Medical Science, The University of Tokyo (IMSUT), established in 1892 as the Institute of Infectious Diseases and renamed IMSUT in 1967, is a leading research institution with a rich history spanning over 130 years. It focuses on exploring biological phenomena and disease principles to develop innovative strategies for disease prevention and treatment. IMSUT fosters a collaborative, interdisciplinary research environment and is known for its work in genomic medicine, regenerative medicine, and advanced medical approaches like gene therapy and AI in healthcare. It operates core research departments and numerous specialized centers, including the Human Genome Center and the Advanced Clinical Research Center, and is recognized as Japan’s only International Joint Usage/Research Center in life sciences.About Dr. Masayuki Yamashita from The Institute of Medical Science, The University of Tokyo, Japan
Dr. Masayuki Yamashita is a physician-scientist specializing in hematology and stem cell biology. He earned his MD from The University of Tokyo in 2008 and his PhD from Keio University School of Medicine in 2014. Following postdoctoral training at UCSF and Columbia University, he joined The University of Tokyo as an Assistant Professor in 2019 and, in 2024, became an Assistant Member at St. Jude Children’s Research Hospital. With over 15 years of research experience and numerous peer-reviewed publications, his work focuses on programmed cell death pathways and hematopoietic stem cell integrity, aiming to develop novel therapies for blood disorders.Funding information
This work was supported by JSPS KAKENHI (23H02707) and the Takeda Science Foundation (Hiroyasu Nakano); JSPS KAKENHI (24H00640, 25H01435, and 25K22629) and AMED (JP20gm1210011 and 25bm1123063) (Keiyo Takubo); and ALSAC, JSPS KAKENHI (20K17395 and 22H03101), the Uehara Memorial Foundation, and Kato Memorial Bioscience Foundation (Masayuki Yamashita). Shared resource core facility support was provided by ALSAC and NIH grant P30CA21765.Media contact
Affiliation: Project Coordination Office, The Institute of Medical Science, The University of Tokyohttps://www.ims.u-tokyo.ac.jp/