Researchers uncover how altered chromatin accessibility reprograms stem cells and fuels disease progression in myelodysplastic syndrome
Myelodysplastic syndrome (MDS) arises from defective blood stem cells that progressively lose their normal functions. Japanese researchers have revealed how changes in chromatin accessibility—how DNA is packaged—reprogram these stem cells toward faulty myeloid gene activity. This shift disrupts the balance of blood cell development and drives disease progression. The team also developed a chromatin-based “progenitor score” that accurately reflects disease severity and predicts patient prognosis in MDS.
Over the past few decades, advances in hematology have illuminated how a delicate balance between stem cell self-renewal and differentiation sustains healthy blood formation. In myelodysplastic syndrome (MDS), however, this balance collapses, leading to abnormal blood cell development and a heightened risk of progression to acute myeloid leukemia. Despite major progress in genetics, the molecular events that trigger this transformation within stem cells have remained unclear.
To address this, a research team led by Professor Atsushi Iwama and Senior Assistant Professor Motohiko Oshima from the Division of Stem Cell and Molecular Medicine, Center for Stem Cell Biology and Regenerative Medicine, The Institute of Medical Science, The University of Tokyo, Japan, has uncovered how chromatin accessibility, the way DNA is packaged and exposed, changes within blood stem cells as MDS develops. The study, published in Volume 16 of the journal Nature Communications on November 28, 2025, provides a new insight into how stem cells lose their normal identity and evolve toward disease.
In healthy bone marrow, hematopoietic stem cells maintain a specific chromatin structure that preserves their renewal potential while suppressing premature activation of differentiation genes. In MDS, the researchers found that this structure is progressively altered. Using the chromatin accessibility and transcriptome analyses, they showed that MDS stem cells gradually lose the hallmarks of normal stem cells and acquire characteristics typical of myeloid progenitors.
This transformation unfolds step by step, reflecting a continuum between healthy and diseased states. “We found that chromatin accessibility in MDS stem cells is remodeled in a direction that primes them for abnormal myeloid differentiation,” explains Prof. Iwama. “These findings reveal a continuum of molecular states between healthy and diseased stem cells, which may be critical to understanding how MDS evolves.”
To measure this process, the team developed a “progenitor score”—a quantitative index based on chromatin accessibility profiles—that tracks how far a cell has moved toward a progenitor-like state. When applied to patient samples, the score correlated strongly with disease severity and prognosis. Patients with higher scores were more likely to experience rapid disease progression or transformation to leukemia.
“Our progenitor cell score can serve as a quantitative indicator of MDS stem cell dysregulation,” says Prof. Iwama. “It could potentially help clinicians identify high-risk patients earlier and tailor their treatment accordingly.”
Beyond diagnostics, the study also highlights how chromatin remodeling—the reshaping of chromatin structure—acts as a driving force in blood cancers. By comparing normal, pre-leukemic, and leukemic stem cells, the researchers found that progressive chromatin changes were a common hallmark of disease evolution. These changes were closely linked to altered transcription factor activity, particularly those governing stem cell maintenance and myeloid differentiation.
This discovery opens the door to new therapeutic possibilities. If chromatin remodeling drives the loss of stem cell identity and the shift toward a transcriptional program, then stabilizing chromatin structure or modulating transcription factor activity could potentially restore normal stem cell function. “Targeting the early epigenetic changes that precede full-blown malignancy may allow us to intervene before the disease becomes aggressive,” notes Dr. Oshima.
This work represents one of the most comprehensive analyses of chromatin accessibility in MDS to date. By integrating epigenomic and transcriptomic data from a large number of patient-derived cells, the team reconstructed the molecular trajectory of MDS stem cells with unprecedented precision.
Ultimately, these results deepen the understanding of how stem cell dysregulation fuels blood disorders and highlight chromatin-based biomarkers as powerful tools for early diagnosis and risk assessment. As chromatin research advances, such approaches may lead to more precise, personalized treatments that prevent disease progression and improve outcomes for patients with MDS.
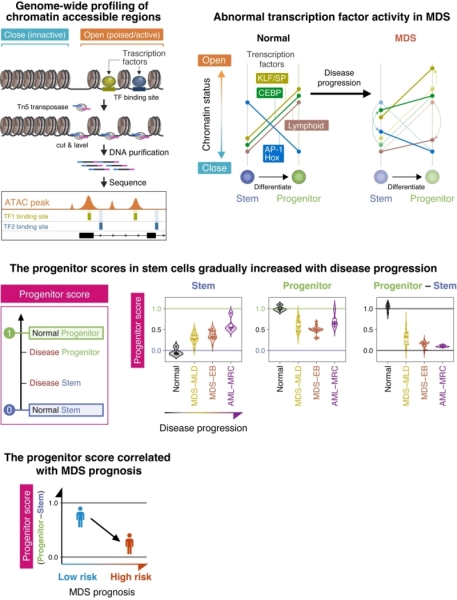
Genome-wide chromatin accessibility profiling reveals how myelodysplastic syndrome (MDS) stem cells progressively lose normal stem-cell features and acquire myeloid progenitor-like characteristics. These chromatin shifts help map the molecular trajectory of disease progression and form the basis of a chromatin-derived “progenitor score” that reflects MDS severity.
Reference
Journal:Nature Communications
Title of original paper:
Chromatin accessibility in stem cells unveils progressive transcriptional alterations in myelodysplastic syndrome
DOI:
10.1038/s41467-025-65753-5
Authors:
Motohiko Oshima1, Naoya Takayama2, Yaeko Nakajima-Takagi1, Daisuke Shinoda1, Naoki Itokawa1, Shuhei Kurosawa1, Satoshi Kaito1, Takahiro Kamiya1, Yuta Yamada1, Shohei Andoh1, Kensuke Kayamori1,3, Sudip Kumar Paul2, Maria Alejandra Kanashiro2, Tomoya Muto3, Shokichi Tsukamoto3, Emiko Sakaida3, Eriko Sato4, Nozomi Yusa5, Kazuaki Yokoyama5, Yasuhito Nannya5, Seiya Imoto6, Bahityar Rahmutulla7, Atsushi Kaneda7,8, Kiyoshi Yamaguchi9, Yoichi Furukawa9, Noriko Doki10, Koji Eto2, Kei Nishikawa11, Ye Ding11, Tomohiro Myojo11, Yuka Harada12, Hironori Harada13, and Atsushi Iwama1,14
Affiliations:
1Division of Stem Cell and Molecular Medicine, Center for Stem Cell Biology and Regenerative Medicine, The Institute of Medical Science, The University of Tokyo, Japan
2Department of Regenerative Medicine, Graduate School of Medicine, Chiba University, Japan
3Department of Hematology, Chiba University Hospital, Japan
4Department of Hematology, Juntendo University Nerima Hospital, Japan
5Department of Hematology/Oncology, IMSUT Hospital, The Institute of Medical Science, The University of Tokyo, Japan
6Division of Health Medical Intelligence, Human Genome Center, The Institute of Medical Science, The University of Tokyo, Japan
7Department of Molecular Oncology, Graduate School of Medicine, Chiba University, Japan
8Health and Disease Omics Center, Chiba University, Japan
9Division of Clinical Genome Research, Advanced Clinical Research Center, The Institute of Medical Science, The University of Tokyo, Japan
10Hematology Division, Tokyo Metropolitan Cancer and Infectious Diseases Center, Komagome Hospital, Japan
11Department of Oncology and Hematology, Edogawa Hospital, Japan
12Department of Clinical Laboratory, Tokyo Metropolitan Cancer and Infectious Diseases Center, Komagome Hospital, Japan
13Laboratory of Oncology, School of Life Sciences, Tokyo University of Pharmacy and Life Sciences, Japan
14Laboratory of Cellular and Molecular Chemistry, Graduate School of Pharmaceutical Sciences, The University of Tokyo, Japan
2Department of Regenerative Medicine, Graduate School of Medicine, Chiba University, Japan
3Department of Hematology, Chiba University Hospital, Japan
4Department of Hematology, Juntendo University Nerima Hospital, Japan
5Department of Hematology/Oncology, IMSUT Hospital, The Institute of Medical Science, The University of Tokyo, Japan
6Division of Health Medical Intelligence, Human Genome Center, The Institute of Medical Science, The University of Tokyo, Japan
7Department of Molecular Oncology, Graduate School of Medicine, Chiba University, Japan
8Health and Disease Omics Center, Chiba University, Japan
9Division of Clinical Genome Research, Advanced Clinical Research Center, The Institute of Medical Science, The University of Tokyo, Japan
10Hematology Division, Tokyo Metropolitan Cancer and Infectious Diseases Center, Komagome Hospital, Japan
11Department of Oncology and Hematology, Edogawa Hospital, Japan
12Department of Clinical Laboratory, Tokyo Metropolitan Cancer and Infectious Diseases Center, Komagome Hospital, Japan
13Laboratory of Oncology, School of Life Sciences, Tokyo University of Pharmacy and Life Sciences, Japan
14Laboratory of Cellular and Molecular Chemistry, Graduate School of Pharmaceutical Sciences, The University of Tokyo, Japan
About The Institute of Medical Science, The University of Tokyo
The Institute of Medical Science, The University of Tokyo (IMSUT), established in 1892 as the Institute of Infectious Diseases and renamed IMSUT in 1967, is a leading research institution with a rich history spanning over 130 years. It focuses on exploring biological phenomena and disease principles to develop innovative strategies for disease prevention and treatment. IMSUT fosters a collaborative, interdisciplinary research environment and is known for its work in genomic medicine, regenerative medicine, and advanced medical approaches like gene therapy and AI in healthcare. It operates core research departments and numerous specialized centers, including the Human Genome Center and the Advanced Clinical Research Center, and is recognized as Japan’s only International Joint Usage/Research Center in life sciences.About Professor Atsushi Iwama from The Institute of Medical Science, The University of Tokyo
Atsushi Iwama, M.D., Ph.D., is a Professor at The Institute of Medical Science, The University of Tokyo, Japan, where he heads the Division of Stem Cell and Molecular Medicine within the Center for Stem Cell Biology and Regenerative Medicine. He earned his doctorate in Medical Science from Kumamoto University, he is renowned for his research on the molecular mechanisms regulating hematopoietic stem cells, leukemia, and aging. Before joining The University of Tokyo, he served at Chiba University’s Graduate School of Medicine. He has published over 300 papers and is an active member of leading international scientific societies.Funding information
This work was supported in part by Grants-in-Aid for 959 Scientific Research (21K08366 and 24K11554 and 19H05653, 19H05746, and 24H00066) from the Japan Society for the Promotion of Science (JSPS) and the Innovative Cancer Medical Practice Research Project (18ck0106401h0001) and Moonshot project (21zf0127003h0001) from AMED, Japan.Media contact
Affiliation: Project Coordination Office, The Institute of Medical Science, The University of Tokyohttps://www.ims.u-tokyo.ac.jp/