Introduction
Our Goal is Triple Fs.
- For the Better Understanding of Mucosal Immunity
- For the Development of Mucosal Vaccine and Immunetherapy
- For the Control of Infectious and Immunological Diseases
Research Projects
MucoRice Project
MucoRice Edible Vaccine
We have developed a molecularly uniform rice-based oral cholera vaccine (MucoRice-CTB) by using an overexpression system for modified cholera toxin B-subunit, CTB (N4Q) with RNAi to suppress production of the major rice endogenous storage proteins. To establish MucoRice-CTB for human use, we have developed hygromycin phosphotransferase (HPT) selection marker–free MucoRice-CTB line 51A by using two different Agrobacterium tumefaciens, each carrying a distinct T-DNA for co-transformation. By whole genome sequencing, we showed that the transgenes in this line were inserted into intergenic regions in chromosome 3 and chromosome 12. For clinical trials, we recently established a prototype of a closed MucoRice hydroponic factory at the Institute of Medical Science, the University of Tokyo, Japan, which was approved as GMP (Good Manufacturing Practices) factory by the Japanese Ministry of Health, Labour and Welfare in 2014. We are now preparing to proceed to clinical trial phase I study of MucoRice-CTB at the Research Hospital at Medical Science Institute, University of Tokyo in 2015.
Oral Antibody Therapy for Gut Infectious Disease
Rotavirus- or norovirus induced diarrhea is a life-threatening disease in children in developing countries and in immunocompromised individuals. We have developed a novel system for prophylaxis and therapy against rotavirus disease using transgenic rice expressing the neutralizing variable domain of a rotavirus-specific llama heavy-chain antibody fragment (MucoRice-ARP1). MucoRice-ARP1 was produced at high levels in rice seeds using an overexpression system and RNAi technology to suppress the production of major rice endogenous storage proteins. Orally administered MucoRice-ARP1 markedly decreased the viral load in immunocompetent as well as immunodeficient mice. MucoRice-ARP1 retained in vitro neutralizing activity after long-term storage and boiling, and conferred protection in mice even after heat treatment at 94 C for 30 min. At present, we are developing orally administered prophylaxis and therapy against norovirus infections.
M-cell Project
The mucosal immune system provides a first line of defense against numerous infectious agents. Intestinal Peyer's patch (PP) and nasopharyngeal associated lymphoreticular tissue (NALT) are considered as a commanding tower for the induction and regulation of mucosal immune responses. PP and NALT possesses a unique antigen sampling cell population "M cells." M cells form an apparent pocket at the basal membrance site where T cells, B cells, macrophages or dendritic cells are situated. It is believed that antigen uptake by M cells does not result in the degradation of the antigen, but rather in the delivery of the intact antigen to the underlying antigen-presenting cells such as macrophages and dendritic cells in the pocket. However, limited immunobiological information, including M cell-specific gene and co-responding surface receptors and/or antigens are currently available. The lack of definitive immunobiological information for M cells has posed an obstacle to mucosal immunologists engaged in designing an effective vaccine delivery vehicle that targets vaccine to mucosal inductive sites such as PP and NALT. In addition, M cells may also facilitate dissemination of infectious pathogens such as Salmonella and Listeria that exploit the M cell transport pathway. Thus, a final goal of this project,elucidation of the molecular and cellular characteristics of M cells would not only greatly facilitate the design of effective mucosal vaccine, but would also lead to the discovery of novel targets for developing new mucosal antigen and drug delivery vehicle to combat pathogens and allergens.
Nasal Vaccine Project
New Generation of Nasal Vaccine
We have demonstrated a cationic type of cholesteryl group-bearing pullulan based nanogel containing pneumococcal surface protein A (PspA-nanogel) induced both protective Th2-mediated Ag-specific systemic and mucosal antibody (Ab) responses and Th17 cell-mediated immunity in mice and non-human primates. In this study, we examine whether PspA-nanogel nasal vaccine could induce PspA-specific protective immunity and cytokines-related miRNA expression. We also examine whether PspA-nanogel nasal vaccine could induce PspA-specific immune reponses in aged mice and macaques as preclinical study to go on human application. Using nanogel-based vaccine technology, we develop mucosal vaccines against lifestyle related diseases as well as other infectious diseases such as tuberculosis.
SCAI project: Analysis of how Structural Cells Affect Immunity
Approach for Overcoming Intestinal Allergies and Inflammatory Diseases
Until now, most immunological studies have involved stimulation of single
immune cell populations, such as T cells and dendritic cells. Those kinds
of experiments, together with immune cell co-culture experiments, have
led to the discovery of many factors involved in allergic and inflammatory
diseases. However, our approach is focused on the interaction of immune
cells and structural cells (e.g. stromal cells, fibroblasts).
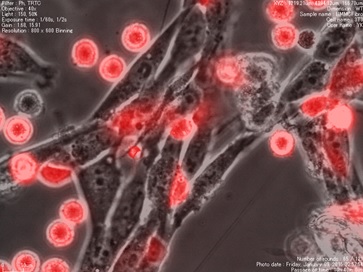
As is shown in the picture, red colored immune cells are tightly attached
to, and communicate with unstained structural cells. We have elucidated
the importance of those communications are important for the acquisition
of “tissue or organ specificity” in immune cells. (Kurashima Y et al., Immunity 2014, Kurashima Y et al., Nat Commun 2012). In addition, we found that once those communications become defective, inflammation takes place U Tokyo Research.
Our project group is now focusing on:
1. Analysis of How “Structural Cells Affect Immunity (SCAI)” using the
CRISPR system
2. Generation of antibodies for allergies and inflammatory disorders in
the mucosa
3. A genomic approach for the treatment of allergies and IBD
4. Generation of an oral immunotherapy model for treatment of food allergies
If you are interested in SCAI, we welcome you to take part in our research
group. Our diverse and friendly group is composed of researchers from various
backgrounds who all share a common enthusiasm for studying immunology.
Our group carries out many exciting and innovative research projects while
maintaining a productive and enjoyable atmosphere. We strongly encourage
you to contact us if our research topics appeal to you.


