Human endogenous retroviruses (HERVs) (*1) have been considered to be junk DNA (*2) in the genome. However, a research team working in the Division of Systems Virology, Department of Infectious Disease Control, IMSUT showed that HERVs function as enhancers in tumors and may contribute to tumor suppression. The results of this research were published in "Science Advances " on October 21, 2020.
Activation of numerous HERVs as enhancers in tumors can changes the characteristics of cancers
HERVs are a type of transposable element that originates from ancient retroviral infections in host germ cells. HERVs have long been regarded as genomic parasites or junk DNA.However, decades of research have shown that HERVs work as enhancers and modulate the expression of host genes. The enhancer activity of HERVs in normal tissues is strictly controlled by epigenetic mechanisms; in contrast, it is known that HERV activity is aberrantly elevated in various types of cancers.
According to the lead scientist, Kei Sato, Associate Professor (Principal Investigator) in IMSUT, it is possible that the activation of numerous HERVs as enhancers in tumors globally alters host gene expression and changes the characteristics of cancers.
To test this hypothesis, the research group of Dr. Sato investigated the multi-omics(*3) dataset of tumors provided by The Cancer Genome Atlas (TCGA)(*4) and assessed the effects of HERV activation on host gene expression.
The research group found that HERV activation in tumors leads to the upregulation of hundreds of transcriptional suppressors, namely KRAB zinc-finger family proteins (KZFPs)(*5). HERVs in the vicinity of KZFP genes work as enhancers in tumors, upregulating the expression of nearby KZFPs.
Highly heterogeneous among patients but strongly associated with disease condition
The research group also found that the activity of HERVs and KZFPs in tumors is highly heterogeneous among patients but strongly associated with disease condition: higher activity of HERV/KZFPs in tumors was associated with milder disease. This result suggests the possibility that the increased expression of KZFPs exerts suppressive effects on tumor progression.Since KZFPs are potent transcriptional suppressors, it is possible that the synchronized induction of many KZFPs in tumors alters gene expression globally and changes the characteristics of tumors.
To test this possibility, the research group assessed the effects of elevated KZFP expression on gene expression patterns and the characteristics of cancer cells. They found that increased KZFP expression in cancer cells suppressed the expression of genes related to cancer progression and suppressed cellular growth, migration, and invasion abilities, suggesting that the increased expression of KZFPs exerts suppressive effects on cancers by modulating the expression of cancer-related genes. Altogether, the present study suggests that HERV activation in tumors drives the synchronized elevation of KZFP expression, presumably leading to tumor suppression (Figure 1).
Associate Professor Sato emphasized the work’s importance; “Gene expression in tumors is highly diverse among patients, but it remains unclear what generates such diversity. We showed that heterogeneity in the activity of the HERV-KZFP gene regulatory network would be one of the causes of the gene expression diversity in tumors. Furthermore, our data suggest that this HERV-KZFP network may orchestrate gene expression for tumor suppression.”
This study was supported in part by JSPS KAKENHI Scientific Research B JP18H02662 (to Kei Sato); JSPS KAKENHI Scientific Research on Innovative Areas JP16H06429 (to Kei Sato), JP16K21723 (to Kei Sato), JP17H05813 (to Kei Sato), and JP19H04826 (to Kei Sato); JSPS KAKENHI Grant-in-Aid for Early-Career Scientists JP20K15767 (to Jumpei Ito); JSPS Research Fellow PD JP19J01713 (to Jumpei Ito) and DC1 JP19J20488 (to Izumi Kimura); The super-computing resource, SHIROKANE, was provided by Human Genome Center, The Institute of Medical Science, The University of Tokyo, Japan.
Figure 1 : Summary of this study
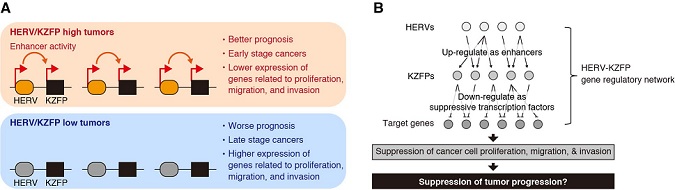
B) The HERV-KZFP gene regulatory network and its proposed function in tumors. Activation of HERVs in tumors induces the coordinated expression of multiple KZFP genes. Increased expression of KZFPs alters the expression of genes related to tumor progression. As a result, the proliferation, migration, and invasion activities of cancer cells are suppressed, presumably resulting in suppression of tumor progression.
Research Notes
(*1) Human endogenous retroviruses (HERVs)A type of transposable element that originated from ancient retroviral infection. In total, 8% of the human genome is composed of HERVs. HERVs have long been regarded as genomic parasites or junk DNA. However, recent studies have revealed that some HERVs work as host genes or transcriptional regulatory sequences such as promoters or enhancers and play critical roles in diverse physiological functions of the host.
(*2) Junk DNA
DNA sequences that are neither genes nor functional elements in the genome. Junk DNA includes transposable elements (a.k.a., mobile genomic elements), simple repeat sequences, and pseudogenes. Junk DNA occupies approximately half of the human genome.
(*3) Multi-omics
An integrated approach to analyzing data sets spanning multiple "-omes", such as the genome, epigenome, transcriptome, proteome, and metabolome.
(*4) The Cancer Genome Atlas (TCGA)
A landmark cancer genomics program in the US, in which over 20,000 primary cancer and matched normal samples spanning 33 cancer types were characterized at the molecular level.
(*5) KRAB zinc-finger family proteins (KZFPs)
The largest family of suppressive transcription factors in higher vertebrates. The human genome encodes approximately 400 KZFP genes. KZFPs bind to their target genes and suppress their expression.
About the research
- 1) Journal Article
Jumpei Ito, Izumi Kimura, Andrew Soper, Alexandre Coudray, Yoshio Koyanagi, Hirofumi Nakaoka, Ituro Inoue, Priscilla Turelli, Didier Trono, and Kei Sato, 2020, “Endogenous retroviruses drive KRAB zinc-finger protein family expression for tumor suppression”, Science Advances.
DOI:https://doi.org/10.1126/sciadv.abc3020
- 2) Publication Journal
Science Advances
https://advances.sciencemag.org/