Humans are exposed continuously to the menace of viral diseases such as those caused by the Ebola virus, Zika virus and coronaviruses. Such emerging/re-emerging viral outbreaks can be triggered by cross-species viral transmission from wild animals to humans.
To achieve cross-species transmission, new hosts have to be exposed to the virus from the old host. Next, the viruses acquire certain mutations that can be beneficial for replicating in the new hosts. Finally, through sustained transmission in the new host, the viruses adapt further evolving as a new virus in the new host (Figure 1). However, at the outset of this process, the viruses have to overcome "the species barriers", which hamper viral cross-species transmission. Mammals including humans have "intrinsic immunity" mechanisms that have diverged enough in evolution to erect species barriers to viral transmission.
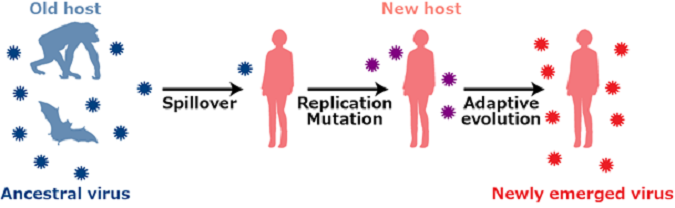
Figure 1:Scheme of viral cross-species transmission
HIV-1 most likely originated from related precursors found in chimpanzees and gorillas
HIV-1, the causative agent of AIDS, most likely originated from related precursors found in chimpanzees (SIVcpz) and gorillas (SIVgor), approximately 100 years ago (Figure 2).
Additionally, SIVgor most likely emerged through the cross-species jump of SIVcpz from chimpanzees to gorillas (Figure 2).
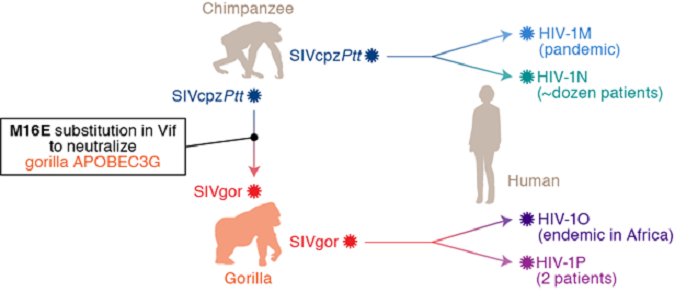
Figure 2 Cross-species transmission of primate lentiviruses leading to the emergence of HIV
However, it remains unclear how primate lentiviruses successfully transmitted among different species. To limit cross-species lentiviral transmission, cellular "intrinsic immunity", including APOBEC3 proteins potentially inhibit lentiviral replication. In contrast, primate lentiviruses in this evolutionary “arms race” have acquired their own "weapon", viral infectivity factor (Vif), to antagonize the antiviral effect of restriction factors.
Additionally, SIVgor most likely emerged through the cross-species jump of SIVcpz from chimpanzees to gorillas (Figure 2).

Figure 2 Cross-species transmission of primate lentiviruses leading to the emergence of HIV
However, it remains unclear how primate lentiviruses successfully transmitted among different species. To limit cross-species lentiviral transmission, cellular "intrinsic immunity", including APOBEC3 proteins potentially inhibit lentiviral replication. In contrast, primate lentiviruses in this evolutionary “arms race” have acquired their own "weapon", viral infectivity factor (Vif), to antagonize the antiviral effect of restriction factors.
Suggesting that a great ape APOBEC3 protein can potentially restrict the cross-species transmission of great ape lentiviruses
A research group at The Institute of Medical Science, The University of Tokyo (IMSUT) showed that gorilla APOBEC3G potentially plays a role in inhibiting SIVcpz replication. Intriguingly, the research group demonstrated that an amino acid substitution in SIVcpz Vif, M16E, is sufficient to overcome gorilla APOBEC3G-mediated restriction.
"To our knowledge, this is the first report suggesting that a great ape APOBEC3 protein can potentially restrict the cross-species transmission of great ape lentiviruses and how lentiviruses overcame this species barrier. Moreover, this is the first investigation elucidating the molecular mechanism by which great ape lentiviruses achieve cross-species transmission", said the lead scientist, Kei Sato, Associate Professor (Principal Investigator) in the Division of Systems Virology, Department of Infectious Disease Control, IMSUT.
"To our knowledge, this is the first report suggesting that a great ape APOBEC3 protein can potentially restrict the cross-species transmission of great ape lentiviruses and how lentiviruses overcame this species barrier. Moreover, this is the first investigation elucidating the molecular mechanism by which great ape lentiviruses achieve cross-species transmission", said the lead scientist, Kei Sato, Associate Professor (Principal Investigator) in the Division of Systems Virology, Department of Infectious Disease Control, IMSUT.
Funding
This study was supported in part by AMED J-PRIDE 19fm0208006 (to Kei Sato); AMED Research Program on HIV/AIDS 19fk0410019 (to Kei Sato) and 19fk0410014 (to Yoshio Koyanagi and Kei Sato); AMED Research Program on Emerging and Re-emerging Infectious Diseases 20fk0108146 (to Kei Sato), 19fk010817 (to So Nakagawa and Kei Sato), and 20fk0108270 (to Yoshio Koyanagi and Kei Sato); JST CREST (to Kei Sato); KAKENHI Grant-in-Aid for Scientific Research B 18H02662 (to Kei Sato), KAKENHI Grant-in-Aid for Scientific Research on Innovative Areas 16H06429 (to So Nakagawa and Kei Sato) KAKENHI Early-Career Scientists 20K15767 (to Jumpei Ito.), and Fund for the Promotion of Joint International Research (Fostering Joint International Research) 18KK0447 (to Kei Sato); JSPS Research Fellow DC1 19J22914 (to Yoriyuki Konno), DC1 19J20488 (to Izumi Kimura), DC1 19J22802 (to Shumpei Nagaoka), 20J23299 (to Hirofumi Aso), and PD 19J01713 (to Jumpei Ito).
About the research
1) Journal Article
Yusuke Nakano, Keisuke Yamamoto, Mahoko Takahashi Ueda, Andrew Soper, Yoriyuki Konno, Izumi Kimura, Keiya Uriu, Ryuichi Kumata, Hirofumi Aso, Naoko Misawa, Shumpei Nagaoka, Soma Shimizu, Keito Mitsumune, Yusuke Kosugi, Guillermo Juarez-Fernandez, Jumpei Ito, So Nakagawa, Terumasa Ikeda, Yoshio Koyanagi, Reuben S Harris and Kei Sato*,2020,“A role for gorilla APOBEC3G in shaping lentivirus evolution including transmission to humans”PLOS Pathogens.
DOI: 10.1371/journal. ppat.1008812
2) Publication Journal
PLOS Pathogens
https://journals.plos.org/plospathogens/
https://journals.plos.org/plospathogens/